Giúp mình câu 5,6 với ạ
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Câu 6 :
$a) C_xH_y + (x + \dfrac{y}{4} ) O_2 \xrightarrow{t^o} xCO_2 + \dfrac{y}{2}H_2O$
$b) 2xFe + yO_2 \xrightarrow{t^o} 2Fe_xO_y$
$c) Fe_xO_y + yCO \xrightarrow{t^o} xFe + yCO_2$
$d) Fe_xO_y + yH_2 \xrightarrow{t^o} xFe + yH_2O$
$e) 2Al + 2NaOH + 2H_2O \to 2NaAlO_2 + 3H_2$
$g) Cu+ 2H_2SO_{4_{đặc}} \xrightarrow{t^o} CuSO_4 +S O_2 + 2H_2O$
$h) 2Fe + 6H_2SO_{4_{đặc}} \xrightarrow{t^o} Fe_2(SO_4)_3 + 3SO_2 + 6H_2O$
Câu 7 :
a)
$Ba +2 H_2O \to Ba(OH)_2 + H_2$
$BaO + H_2O \to Ba(OH)_2$
b)
$n_{Ba} = n_{H_2} = \dfrac{4,48}{22,4} = 0,2(mol)$
$\Rightarrow n_{BaO} = \dfrac{58 - 0,2.137}{153} = 0,2(mol)$
$m_{dd} = 58 + 200 - 0,2.2 = 257,6(gam)$
$C\%_{Ba(OH)_2} = \dfrac{(0,2 + 0,2).171}{257,6}.100\% = 26,55\%$

5a.
Pt có 2 nghiệm pb lhi:
\(\Delta=9+4m>0\Leftrightarrow m>-\dfrac{9}{4}\)
b. Phương trình có 2 nghiệm khi:
\(\Delta=1+4\left(-2m+1\right)\ge0\Rightarrow m\le\dfrac{5}{8}\)
6.
a. Pt có 2 nghiệm khi:
\(\Delta'=1-\left(m+2\right)\ge0\Leftrightarrow m\le-1\)
6b
Khi \(m\le-1\), theo hệ thức Viet ta có: \(\left\{{}\begin{matrix}x_1+x_2=2\\x_1x_2=m+2\end{matrix}\right.\)
\(x^2_1+x^2_2=10\Leftrightarrow\left(x_1+x_2\right)^2-2x_1x_2=10\)
\(\Leftrightarrow4-2\left(m+2\right)=10\)
\(\Leftrightarrow m=-5\)
B.
\(x^2_1+x_2^2+4x_1x_2=0\)
\(\Leftrightarrow\left(x_1+x_2\right)^2+2x_1x_2=0\)
\(\Leftrightarrow4+2\left(m+2\right)=0\)
\(\Leftrightarrow m=-4\)





5. a) Fe + H2SO4 --------> FeSO4 + H2
Mg + H2SO4 --------> MgSO4 + H2
Gọi x, y lần lượt là số mol của Fe, Mg
Ta có \(\left\{{}\begin{matrix}56x+24y=12\\x+y=\dfrac{6,72}{22,4}=0,3\end{matrix}\right.\)
=>\(\left\{{}\begin{matrix}x=0,15\\y=0,15\end{matrix}\right.\)
=> \(\%m_{Fe}=\dfrac{0,15.56}{12}.100=70\%\)
=> %mMg = 100 -70 = 30%
b) Ta có n H2SO4 = n Fe + nMg = 0,15 + 0,15 =0,3 (mol)
=>a = \(CM_{H_2SO_4}=\dfrac{0,3}{0,2}=1,5M\)
c) \(CM_{FeSO_4}=\dfrac{0,15}{0,2}=0,75M\)
\(CM_{MgSO_4}=\dfrac{0,15}{0,2}=0,75M\)
6. a) 2Al + 6HCl → 2AlCl3 + 3H2
Zn + 2HCl →ZnCl2 + H2
Gọi x, y lần lượt là số mol của Al, Zn
\(\left\{{}\begin{matrix}27x+65y=11,9\\\dfrac{3}{2}x+y=\dfrac{8,96}{22,4}\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}x=0,2\\y=0,1\end{matrix}\right.\)
=> \(\%m_{Al}=\dfrac{27.0,2}{11,9}.100=45,38\%\)
=> %m Zn = 100- 45,38=54,62%
b) \(a=C\%_{HCl}=\dfrac{\left(0,2.2+0,1.2\right).36,5}{200}.100=10,95\%\)
c) \(m_{ddsaupu}=11,9+200-\left(0,2.2+0,1.2\right)=211,3\left(g\right)\)
=> \(C\%_{AlCl_3}=\dfrac{0,2.133,5}{211,3}.100=12,64\%\)
\(C\%_{ZnCl_2}=\dfrac{0,1.136}{211,3}.100=6,44\%\)




 Giúp câu 5,6 tự luận với ạ
Giúp câu 5,6 tự luận với ạ

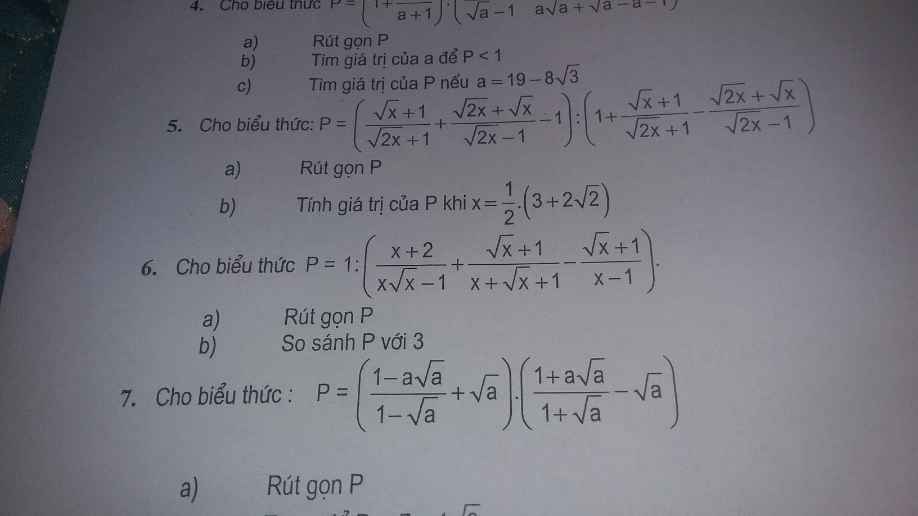

Câu 5:
PTHH: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
a_____3a______a______ \(\dfrac{3}{2}a\) (mol)
\(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\)
b_____2b_____b______b (mol)
a) Ta lập được hệ phương trình: \(\left\{{}\begin{matrix}27a+56b=5,5\\\dfrac{3}{2}a+b=\dfrac{4,48}{22,4}=0,2\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}a=0,1\\b=0,05\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Al}=\dfrac{0,1\cdot27}{5,5}\cdot100\%\approx49,1\%\\\%m_{Fe}=50,9\%\end{matrix}\right.\)
b) Theo các PTHH: \(n_{HCl}=3n_{Al}+2n_{Fe}=0,4\left(mol\right)\) \(\Rightarrow V_{ddHCl}=\dfrac{0,4}{0,5}=0,8\left(l\right)\)
c) Theo PTHH: \(\left\{{}\begin{matrix}n_{AlCl_3}=0,1\left(mol\right)\\n_{FeCl_2}=0,05\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}C_{M_{AlCl_3}}=\dfrac{0,1}{0,8}=0,125\left(M\right)\\C_{M_{FeCl_2}}=\dfrac{0,05}{0,8}=0,0625\left(M\right)\end{matrix}\right.\)
Câu 6:
a+b) Ta có: \(\left\{{}\begin{matrix}\Sigma n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\\n_{Zn}=\dfrac{9,75}{65}=0,15\left(mol\right)\end{matrix}\right.\)
PTHH: \(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\uparrow\)
0,15___0,15_____0,15___0,15 (mol)
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\uparrow\)
0,25___0,25____0,25____0,25 (mol)
\(\Rightarrow m_{Fe}=0,25\cdot56=14\left(g\right)\)
c) PTHH: \(CuO+H_2\xrightarrow[]{t^o}Cu+H_2O\)
Ta có: \(\left\{{}\begin{matrix}n_{H_2}=0,4\left(mol\right)\\n_{CuO}=\dfrac{24}{80}=0,3\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\) H2 còn dư, CuO p/ứ hết
\(\Rightarrow n_{Cu}=0,3\left(mol\right)\) \(\Rightarrow m_{Cu}=0,3\cdot64=19,2\left(g\right)\)