Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a,\(n_{H_2SO_4}=0,5.0,2=0,1\left(mol\right)\)
PTHH: H2SO4 + 2KOH → K2SO4 + 2H2O
Mol: 0,1 0,2 0,1
b,\(C_{M_{ddKOH}}=\dfrac{0,2}{0,05}=4M\)
c,Vdd sau pứ = 0,2+0,05 = 0,25 (l)
\(C_{M_{ddK_2SO_4}}=\dfrac{0,1}{0,25}=0,4M\)

\(n_{H_2}=\dfrac{6,72}{22,4}=0,3mol\)
\(\left\{{}\begin{matrix}n_{Mg}=x\left(mol\right)\\n_{Zn}=y\left(mol\right)\end{matrix}\right.\Rightarrow24x+65y=11,3\left(1\right)\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(\Rightarrow x+y=0,3\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}x=0,2mol\\y=0,1mol\end{matrix}\right.\)
a)\(\%m_{Mg}=\dfrac{0,2\cdot24}{11,3}\cdot100\%=42,48\%\)
\(\%m_{Zn}=100\%-42,48\%=57,52\%\)
b)\(n_{HCl}=2\left(n_{Mg}+n_{Zn}\right)=2\cdot\left(0,2+0,1\right)=0,6mol\)
\(C_{M_{HCl}}=\dfrac{0,6}{0,2}=3M\)

Có \(m_M=m_{Na}+m_{Cl}+m_{SO_4}=1,38+m_{Cl}+m_{SO_4}=3,76\)
\(\Rightarrow m_{Cl}+m_{SO_4}=2,38\)
\(\Rightarrow35,5n_{HCl}+96n_{H_2SO_4}=2,38\)
Lại có : \(n_O=n_{NaOH}=0,06\left(mol\right)\)
BtH : \(n_H=2n_{H_2O}=2n_O=n_{H\left(NaOH\right)}+n_{H\left(HCl\right)}+2n_{H\left(H_2SO_4\right)}\)
\(\Rightarrow n_{HCl}+2n_{H_2SO_4}=0,06\)
\(\Rightarrow n_{HCl}=0,04\left(mol\right)\)
\(\Rightarrow C_{MHCl}=1M\)

Chọn đáp án C
n C l 2 = 1 , 12 22 , 4 = 0,05 (mol); n N a O H d ư = 0,2.0,5 = 0,1 (mol)
C l 2 + 2NaOH → NaCl + NaClO + H 2 O
0,05 0,1 (mol)
=> n N a O H b đ = 0,1 + 0,1 = 0,2 (mol) => C M N a O H = = 1,0 (M)

\(a) n_{Fe_2O_3} = \dfrac{8}{160} = 0,05(mol)\\ Fe_2O_3 + 6HCl \to 2FeCl_3 + 3H_2O\\ n_{FeCl_3} = 2n_{Fe_2O_3} = 0,1(mol)\\ m_{FeCl_3} = 0,1.162,5 = 16,25(gam)\\ b) n_{HCl} = 6n_{Fe_2O_3} = 0,05.6 = 0,3(mol)\\ V_{dd\ HCl} = \dfrac{0,3}{0,5} = 0,6(lít)\\ c) C_{M_{FeCl_3}} = \dfrac{0,1}{0,5} = 0,2M\)

\(n_{Mg}=\dfrac{4,8}{24}=0,2\left(mol\right)\\ a.Mg+2HCl\rightarrow MgCl_2+H_2\\ n_{H_2}=n_{MgCl_2}=n_{Mg}=0,2\left(mol\right)\\ V_{H_2\left(đktc\right)}=0,2.22,4=4,48\left(l\right)\\ b.n_{HCl}=0,2.2=0,4\left(mol\right)\\ m_{ddHCl}=\dfrac{0,4.36,5.100}{7,3}=200\left(g\right)\\ c.m_{ddsau}=4,8+200-0,2.2=204,4\left(g\right)\\ C\%_{ddMgCl_2}=\dfrac{0,2.95}{204,4}.100\approx9,295\%\\ d.V_{ddHCl}=\dfrac{200}{1,05}=\dfrac{4000}{21}\left(ml\right)=\dfrac{4}{21}\left(l\right)\\ C_{MddHCl}=\dfrac{0,4}{\dfrac{4}{21}}=2,1\left(M\right)\)
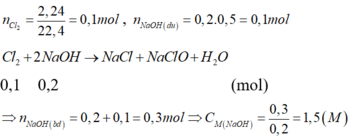
a,\(n_{H_2SO_4}=0,5.0,2=0,1\left(mol\right)\)
PTHH: H2SO4 + 2KOH → K2SO4 + H2O
Mol: 0,1 0,2 0,2
b,\(C_{M_{ddKOH}}=\dfrac{0,2}{0,05}=4M\)
c, Vdd sau pứ = 0,2+0,05 = 0,25 (l)
\(C_{M_{ddK_2SO_4}}=\dfrac{0,1}{0,25}=0,4M\)