Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{P_2O_5}=\dfrac{7,1}{142}=0,05mol\)
\(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
0,1 0,125 0,05
\(V_{O_2}=0,125\cdot22,4=2,8l\)
\(m_P=0,1\cdot31=3,1g\)
\(n_{P_2O_5}=\dfrac{7,1}{142}=0,05mol\)
\(4P+5O_2\rightarrow\left(t^o\right)2P_2O_5\)
0,1 0,125 0,05 ( mol )
\(V_{O_2}=0,125.22,4=2,8l\)
\(m_P=0,1.31=3,1g\)

a)\(n_{Al}=\dfrac{5,4}{27}=0,2\left(m\right)\)
\(PTHH:4Al+3O_2\underrightarrow{t^o}2Al_2O_3\)
tỉ lệ :4 3 2
số mol :0,2 0,15 0,1
\(V_{O_2}=0,15.22,4=3,36\left(l\right)\)
b)\(m_{Al_2O_3}=0,1.102=10,2\left(g\right)\)
c)\(PTHH:2KMnO_4\underrightarrow{t^o}K_2MnO_4+MnO_2+O_2\)
tỉ lệ :2 1 1 1
số mol :0,3 0,15 0,15 0,15
\(m_{KMnO_4}=0,3.126=37,8\left(g\right)\)

4P + 5O2 ----> 2P2O5
0,24 -> 0,3 ---> 0,12 (mol)
nP = \(\dfrac{7,44}{31}\)= 0,24 (mol)
VH2 = 0,3 . 22,4 = 6,72 (l)
2KClO3 ---> 2KCl + 3O2
0,2 <------------- 0,3 (mol)
mKClO3 = 0,2 . (39 + 35,5 + 16.3)
= 24,5 (g)
Vui lòng kiểm tra lại kết quả dùm, thank you.
nP = 7,44 : 31 = 0,24 ( mol)
pthh : 4P + 5O2 -t--> 2P2O5
0,24->0,3 (mol)
=> VO2 =0,3 . 22,4 = 6,72 (l)
pthh : 2KClO3 -t--> 2KCl + 3O2
0,2<-------------------0,3 (mol)
=> mKClO3 = 0,2 .122,5 = 24,5 (g)

a)\(n_{Fe}=\dfrac{22,4}{56}=0,4mol\)
\(3Fe+2O_2\underrightarrow{t^o}Fe_3O_4\)
0,4 \(\dfrac{4}{15}\) \(\dfrac{2}{15}\)
\(V_{O_2}=\dfrac{4}{15}\cdot22,4=5,973l\)
b)\(2KClO_3\underrightarrow{t^o}2KCl+3O_2\)
\(\dfrac{8}{45}\) \(\dfrac{4}{15}\)
\(m_{KClO_3}=\dfrac{8}{45}\cdot122,5=21,78g\)

a, \(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
b, \(n_P=\dfrac{12,4}{31}=0,4\left(mol\right)\)
Theo PT: \(n_{P_2O_5}=\dfrac{1}{2}n_P=0,2\left(mol\right)\Rightarrow m_{P_2O_5}=0,2.142=28,4\left(g\right)\)
c, \(n_{O_2}=\dfrac{5}{4}n_P=0,5\left(mol\right)\)
\(\Rightarrow V_{O_2}=0,5.22,4=11,2\left(l\right)\)
d, \(2KClO_3\underrightarrow{t^o}2KCl+3O_2\)
Theo PT: \(n_{KClO_3}=\dfrac{2}{3}n_{O_2}=\dfrac{1}{3}\left(mol\right)\Rightarrow m_{KClO_3}=\dfrac{1}{3}.122,5=\dfrac{245}{6}\left(g\right)\)

\(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\\ a,2Mg+O_2\rightarrow\left(t^o\right)2MgO\\ n_{O_2}=\dfrac{0,1}{2}=0,05\left(mol\right)\\ V_{O_2\left(đktc\right)}=0,05.22,4=1,12\left(l\right)\\ b,2KClO_3\rightarrow\left(t^o\right)2KCl+3O_2\\ n_{KClO_3}=\dfrac{0,05.2}{3}=\dfrac{1}{30}\left(mol\right)\\ \Rightarrow m_{KClO_3}=\dfrac{122,5}{30}=\dfrac{49}{12}\left(g\right)\)

\(n_P=\dfrac{12,4}{31}=0,4\left(mol\right)\\ a,PTHH:4P+5O_2\rightarrow\left(t^o\right)2P_2O_5\\ b,n_{O_2}=\dfrac{5}{4}.0,4=0,5\left(mol\right)\\ V_{O_2\left(đktc\right)}=0,5.22,4=11,2\left(l\right)\\ c,n_{P_2O_5}=\dfrac{2}{4}.0,4=0,2\left(mol\right)\\ m_{P_2O_5}=142.0,2=28,4\left(g\right)\)

nP = 12,4/31 = 0,4 (mol)
PTHH: 4P + 5O2 -> (t°) 2P2O5
Mol: 0,4 ---> 0,5 ---> 0,2
mP2O5 = 0,2 . 142 = 28,4 (g)
VO2 = 0,5 . 22,4 = 11,2 (l)
PTHH: 2KMnO4 -> (t°) K2MnO4 + MnO2 + O2
nKMnO4 = 0,5 . 2 = 1 (mol)
mKMnO4 = 1 . 158 = 158 (g)
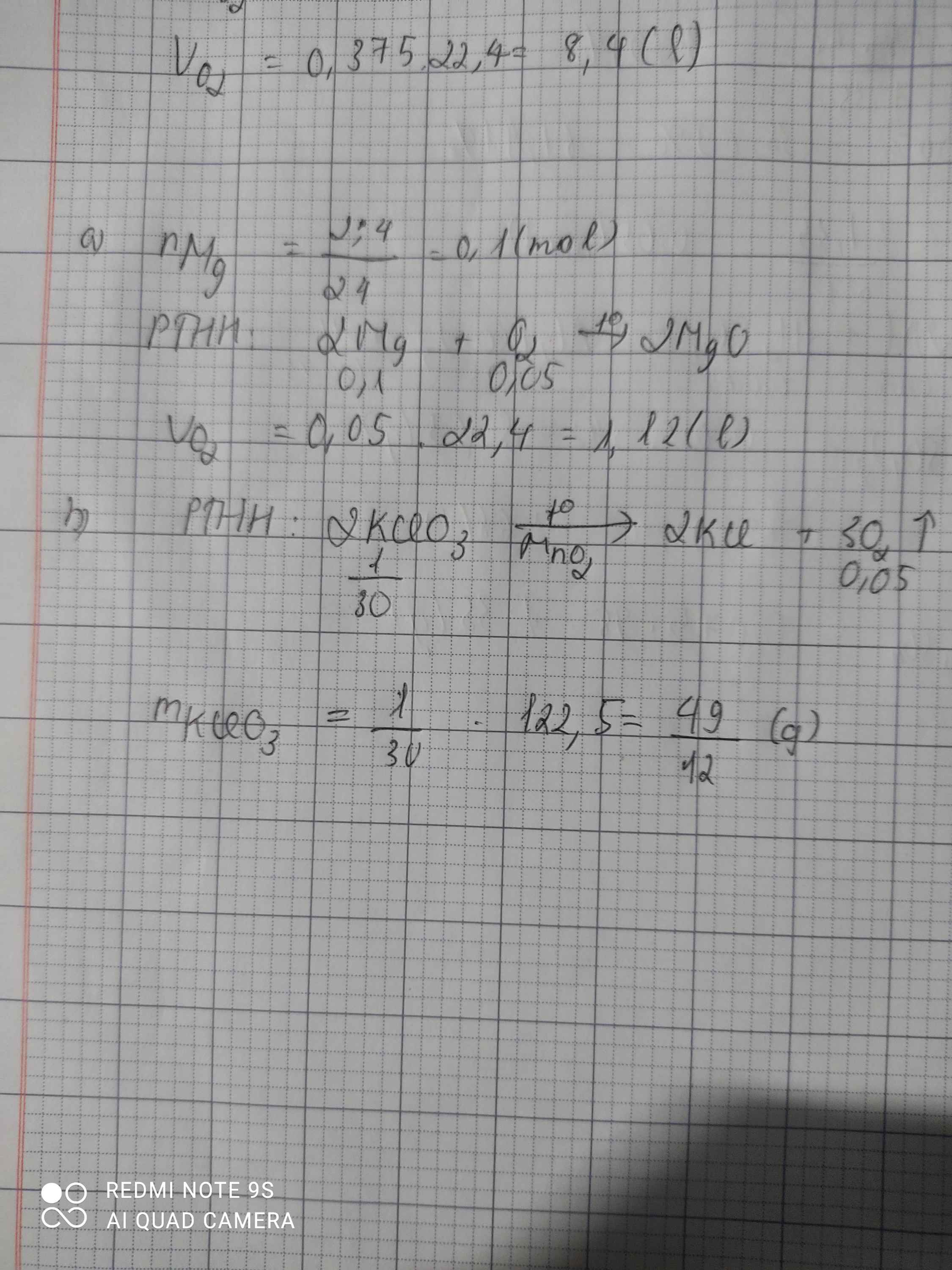

\(n_{P_2O_5}=\dfrac{28.4}{142}=0.2\left(mol\right)\)
\(4P+5O_2\underrightarrow{t^0}2P_2O_5\)
\(.......0.5......0.2\)
\(V_{O_2}=0.5\cdot22.4=11.2\left(l\right)\)
\(2KClO_3\underrightarrow{t^0}2KCl+3O_2\)
\(\dfrac{1}{3}...................0.5\)
\(m_{KClO_3}=\dfrac{1}{3}\cdot122.5=40.83\left(g\right)\)
a) nP2O5= 28,4/142= 0,2(mol)
PTHH: 4P + 5 O2 -to-> 2 P2O5
nO2= 5/2 . 0,2= 0,5(mol)
=>V(O2,đktc)=0,5.22,4= 11,2(l)
b) 2KClO3 -to-> 2 KCl + 3 O2
1/3_________________0,5(mol)
mKClO3= 1/3 . 122,5\(\approx\) 40,833(g)